Abstract
Diagnosis of low-grade myelodysplastic syndromes (LG-MDS) is one of the most challenging in hematopathology as it relies predominantly on morphologic assessment of dysplasia. Prior studies have demonstrated poor interobserver agreement among pathologists. Histomorphological evaluation of bone marrow core biopsy samples remains the gold standard for diagnostic workup of LG-MDS, including myelodysplastic syndromes (MDS) and other myeloid neoplasms. However, this approach may be subjective, and cannot quantitatively assess subtle differences in marrow topography and the cellular microenvironment. Multiparametric in situ imaging (MISI) through various techniques enables multiple biomarker detection in a single tissue. BostonGene has developed an AI-based image analysis platform to reveal spatial information and subtle histomorphologic features in an objective, quantitative fashion. Here, we demonstrate the potential for automated AI-based imaging analysis of MISI to assist in the differentiation of LG-MDS samples from normal marrow tissues (NBM).
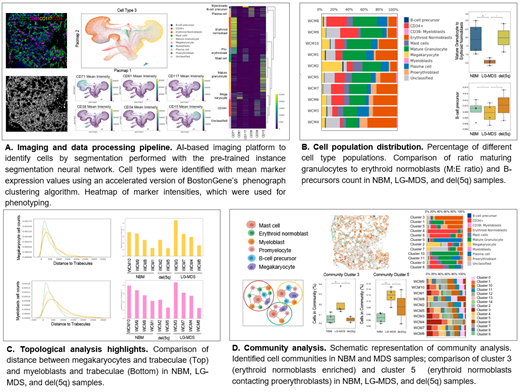
Decalcified human bone marrow core biopsy tissues from LG-MDS (n=6) and uninvolved staging marrows (NBM, n=4) were first prepared by immunofluorescence-based MISI via staining with DAPI and CD34, CD38, CD117, CD71, CD15, and CD61 antibodies. BostonGene analyzed the resulting images (fig.1) using a proprietary AI-based imaging platform to identify cells by segmentation performed with the pre-trained instance segmentation neural network. Cell types were identified with mean marker expression values using an accelerated version of BostonGene's phenograph clustering algorithm. Pathologists manually masked fat and bone trabeculae.
Using a combination of cell size/shape parameters and antigen expression levels, the following unique cell types were identified: myeloblasts, proerythroblasts, erythroid normoblasts, maturing granulocytes, megakaryocytes, mast cells, plasma cells, and B-cell precursors (hematogones). Data revealed differences in the cellular content of NBM and LG-MDS samples, and separation of LG-MDS samples with the del(5q) subtype (n=2). While linear slender islet-like small clusters of erythroid normoblasts were detected in NBM, we observed a chaotic arrangement of them in LG-MDS samples. In LG-MDS samples, we found an increase in the total number of erythroid normoblasts from 17% to 31%, LG-MDS-del(5q) had 14%.
The ratio of maturing granulocytes to erythroid normoblasts (M:E ratio) was significantly lower in LG-MDS (0.63) than in NBM (1.95). The M:E ratio generated by MISI strongly correlated to the M:E ratio produced by manual differential count of bone marrow aspirate samples (R=0.83, p < 0.003). Additionally, fewer hematogones were identified in LG-MDS marrows compared to NBM samples, as reported by others using orthogonal methods.
Topographic analysis showed the fat to cellular tissue area ratio was higher in NBM (0.73) than LG-MDS (0.41), but the ratio of trabecular area to total tissue area was higher in LG-MDS (1.67) than NMB (0.74). Spatially, myeloblasts and megakaryocytes were found closer to trabeculae in NBM than LG-MDS;12 different cell communities were identified;2 of them (cluster 3 - erythroid normoblasts enriched, cluster 5 - erythroid normoblasts contacting proerythroblasts) were distributed statistically significantly differently in NBM and LG-MDS samples, indicating the use of MISI with AI-based imaging to distinguish LG-MDS from NBM. Patients of MDS-del(5q) subtype differ significantly from other MDS samples and are more similar to NBM.
AI-based image analysis applied to MISI of bone marrow tissue revealed multiple cell types in single tissue sections, along with histologically subtle differences in topography between NBM and LG-MDS samples. These results highlight the importance of integrating in situ tissue analysis with techniques that examine single cell characteristics for a more comprehensive picture of the differences between normal tissue and tumor samples. Coupling sophisticated imaging analytics with this imaging method may provide a more powerful tool for novel biomarker discovery of prognostic and therapeutic significance in the management of MDS and other marrow-based disorders.
Svekolkin: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties. Varlamova: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties. Galkin: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties. Akaeva: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties. Smirnova: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties. Ovcharov: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties. Polyakova: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties. Tabakov: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company. Postovalova: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties: BostonGene. Koltakova: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company. Gunn: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company. Bagaev: BostonGene Corp.: Current Employment, Current holder of stock options in a privately-held company, Patents & Royalties: BostonGene.